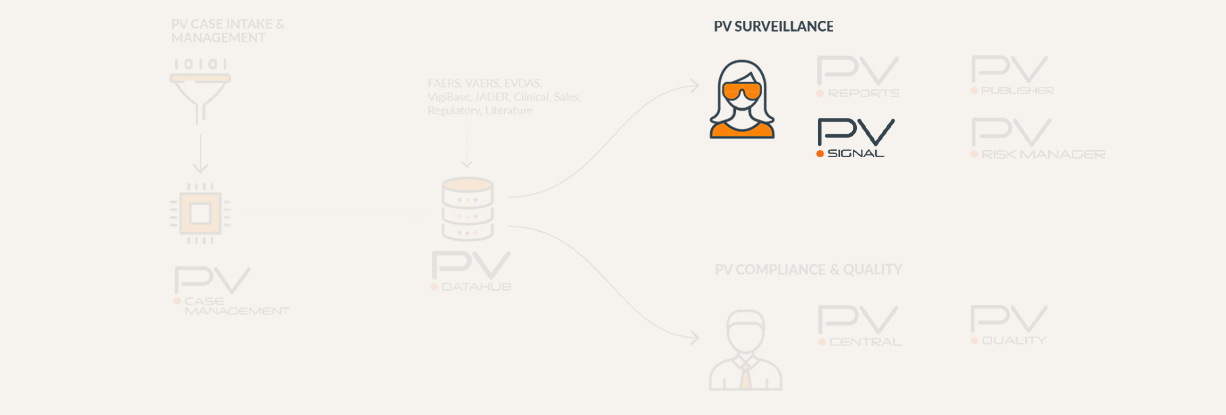
Comprehensive, Compliant and User-Friendly Signal Detection and Management Solution
PV Signal is the industry’s most complete signal detection and management solution. It is fully compliant with EU GPV Module IX regulations. It provides a dynamic data mining environment for detecting signals, uncovering patterns, and recognizing emerging trends in spontaneous adverse event report data. PV Signal supports seamless review across various data sources and provides advanced signal analysis using machine learning and AI based technologies.
Save Operational time by leveraging PV Signal and instead use your time to focus on patient safety, long-term sustainability, and public health.